 /periodic-table-165930186-590f2d703df78c92832fe141.jpg)
Examples: H 2, Cl 2, Br 2, I 2, HCl, NH 3, CH 4, and C 2 H 6. When one pair of electrons, or two electrons, are shared between the atoms, it is known as a single covalent bond or merely a single bond. It's thought that another one of these magic numbers lies just beyond the range of currently known elements, which means we could potentially discover some new elements that are relatively stable and extremely massive. Based on the number of shared electron pairs, there are three types of covalent bonds 1-6: 1. There are believed to be certain numbers/ratios that are inherently more stable. 
Hence, larger atoms become more unstable. A molecule may consist of two or more atoms of a single element or atoms of different elements. The ratio of neutrons to protons gradually increases to try to compensate, but this results in the neutrons getting more unstable themselves, resulting in more opportunity for weak forces to destabilize them. halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table.The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). With all of that to consider, as you get larger and larger nuclei with more protons, the electromagnetic forces increase, as well as the distances in the nuclei, putting more strain on the nuclear forces holding it all together. Periodic Law states that chemical and physical properties of elements. Basically, what this means is if you drop down a row (period) on the table and move across it, elements follow the same trend as other periods. 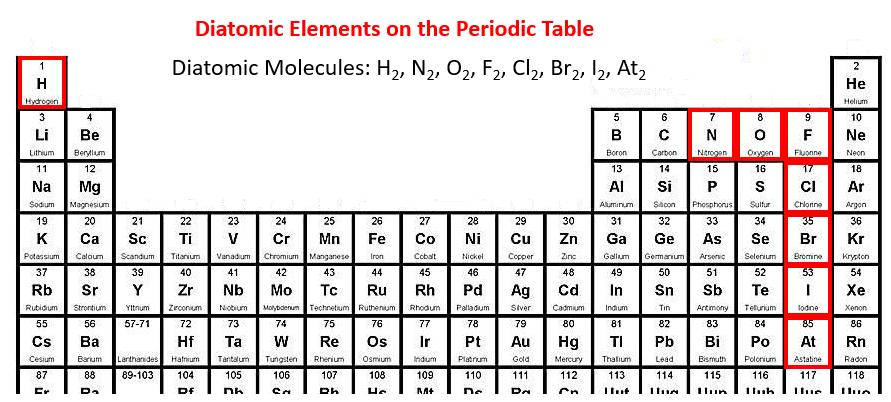
A Brønsted-Lowry acid is any species that can donate a proton (H +) to another molecule. Neutrons will help add to this nuclear force, but they tend to become unstable when they get into too great of concentration and the weak force will cause them to decay into protons. In chemistry, periodicity refers to repeating trends in element properties on the periodic table. In 1923, Danish chemist Johannes Brønsted and English chemist Thomas Lowry independently proposed new definitions for acids and bases, ones that focus on proton transfer. They are generally asymmetrical, with an uneven distribution of the electrons. One end is slightly positive one end is slightly negative. Fusion allows these protons to get close enough together for the stronger, yet short ranged, nuclear force to overpower the electromagnetic force. In chemistry, the definition of a polar molecule, is a molecule that has a charge on one side of the molecule, that is not cancelled out. The electromagnetic force of the protons wants to push them away from each other. A key concept in a discussion of chemical bonding is that of the molecule. There are several forces at work inside the nuclei of atoms. The type of bond that is most likely to occur between two atoms can be predicted on the basis of the location of the elements in the periodic table, and to some extent the properties of the substances so formed can be related to the type of bonding. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
